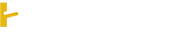Giúp em giải mấy cái dãy chuyển hóa này với ạ
Hãy luôn nhớ cảm ơn và vote 5*
nếu câu trả lời hữu ích nhé!
TRẢ LỜI
- nguyencongdinhdhy

Đáp án:
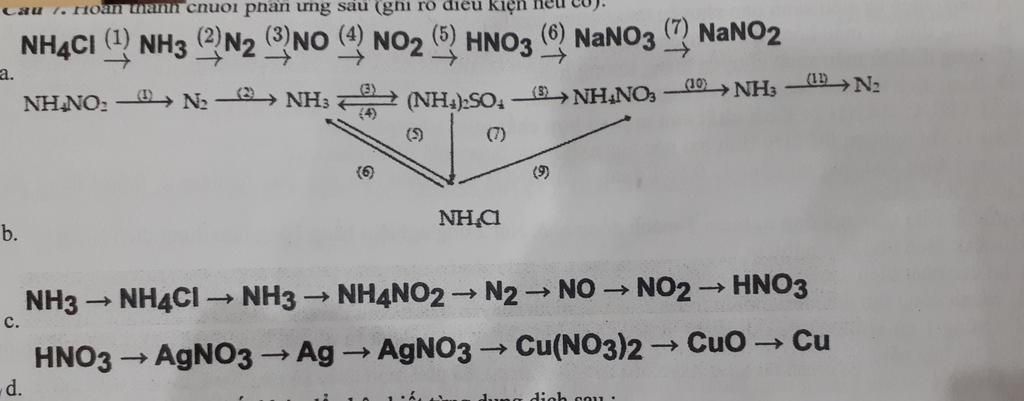
\(a)\)
\(NH_4^{}Cl + NaOH \to NH_3^{} + H_2^{}O + NaCl\left( 1 \right)\)
\(4NH_3^{} + 3O_2^{}\xrightarrow{{{{900}^o}C}}2N_2^{} + 6H_2^{}O\left( 2 \right)\)
\(N_2^{} + O_2^{}\xrightarrow{{{{3000}^o}C}}2NO\left( 3 \right)\)
\(NO + O_2^{}\xrightarrow{{{t^o}}}NO_2^{}\left( 4 \right)\)
\(2NO_2^{} + H_2^{}O + \frac{1}{2}O_2^{} \to 2HNO_3^{}\left( 5 \right)\)
\(HNO_3^{} + NaOH \to NaNO_3^{} + H_2^{}O\left( 6 \right)\)
\(2NaNO_3^{}\xrightarrow{{{t^o}}}2NaNO_2^{} + O_2^{}\left( 7 \right)\)
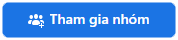
\(b)\)
\(NH_4^{}NO_2^{}\xrightarrow{{{t^o}}}N_2^{} + 2H_2^{}O\)
\(N_2^{} + 3H_2^{}\xrightarrow{{xt,{t^o}}}2NH_3^{}\)
\(2NH_3^{} + H_2^{}SO_4^{} \to (NH_4^{})_2^{}SO_4^{}\)
\((NH_4^{})_2^{}SO_4^{} + Ba(NO_3^{})_2^{} \to 2NH_4^{}NO_3^{} + BaSO_4^{}\)
\(NH_4^{}NO_3^{} + NaOH \to NH_3^{} + NaNO_3^{}\)
\(4NH_3^{} + 3O_2^{}\xrightarrow{{{{900}^o}C}}2N_2^{} + 6H_2^{}O\)
\(NH_3^{} + HCl \to NH_4^{}Cl\left( 5 \right)\)
\(NH_4^{}Cl\xrightarrow{{{t^o}}}NH_3^{} + HCl\left( 6 \right)\)
\((NH_4^{})_2^{}SO_4^{} + BaCl_2^{} \to 2NH_4^{}Cl + BaSO_4^{}\left( 7 \right)\)
\(NH_4^{}Cl + AgNO_3^{} \to AgCl + NH_4^{}NO_3^{}\left( 9 \right)\)
\(c)NH_3^{} + HCl \to NH_4^{}Cl\)
\(NH_4^{}Cl\xrightarrow{{{t^o}}}NH_3^{} + HCl\)
\(NH_3^{} + HNO_2^{} \to NH_4^{}NO_2^{}\)
\(NH_4^{}NO_2^{}\xrightarrow{{{t^o}}}N_2^{} + 2H_2^{}O\)
\(N_2^{} + O_2^{}\xrightarrow{{{{3000}^o}C}}2NO\)
\(NO + O_2^{}\xrightarrow{{{t^o}}}NO_2^{}\)
\(2NO_2^{} + H_2^{}O + \frac{1}{2}O_2^{} \to 2HNO_3^{}\)
\(d)\)
\(4HNO_3^{} + 2Ag \to 2AgNO_3^{} + NO_2^{} + 2H_2^{}O\)
\(AgNO_3^{}\xrightarrow{{{t^o}}}Ag + NO_2^{} + \frac{1}{2}O_2^{}\)
\(4HNO_3^{} + 2Ag \to 2AgNO_3^{} + NO_2^{} + 2H_2^{}O\)
\(AgNO_3^{} + CuCl_2^{} \to Cu(NO_3^{})_2^{} + AgCl\)
\(Cu(NO_3^{})_2^{}\xrightarrow{{{t^o}}}CuO + 2NO_2^{} + O_2^{}\)
\(CuO + H_2^{}\xrightarrow{{{t^o}}}Cu + H_2^{}O\)
Hãy giúp mọi người biết câu trả lời này thế nào?
![]()
Bạn muốn hỏi điều gì?
Bảng tin
Bạn muốn hỏi điều gì?
 Inbox: m.me/hoidap247online
Inbox: m.me/hoidap247online Trụ sở: Tầng 7, Tòa Intracom, số 82 Dịch Vọng Hậu, Cầu Giấy, Hà Nội.
Trụ sở: Tầng 7, Tòa Intracom, số 82 Dịch Vọng Hậu, Cầu Giấy, Hà Nội.